
If boron has an atomic mass of 10.81 amu, which isotope occurs in greater abundance. Boron-10 and boron-11 are the naturally occurring isotopes of elemental boron. Its number of neutrons is the sum of its. So, we have found that boron has 5 protons, 6 neutrons and 5 electrons. Challenge An atom has a mass number of 55. Write the symbols for the two isotopes of boron and determine the relative atomic mass. The atomic masses of Boron-10 and Boron-11 are 10.0129 amu and 11.0093 amu respectively. For boron, we take 5 away from 11, which gives us 6 (11 – 5 = 6). Science Chemistry PRACTICE EXERCISE: Problem Solving Boron has two isotopes, Boron-10 and Boron-11, whose percentage abundances are 19.8 and 80.2 respectively. The atomic number gives us the number of protons, so we can work out the number of neutrons in an element by taking the atomic number (number of protons/ bottom number) away from the mass number (top number). Boron has a mass number of 11 the number of protons and neutrons in boron is 11. number of elements after they are slowed by passage through a hydrogen - rich. The isotope boron-10 is good at absorbing neutrons. 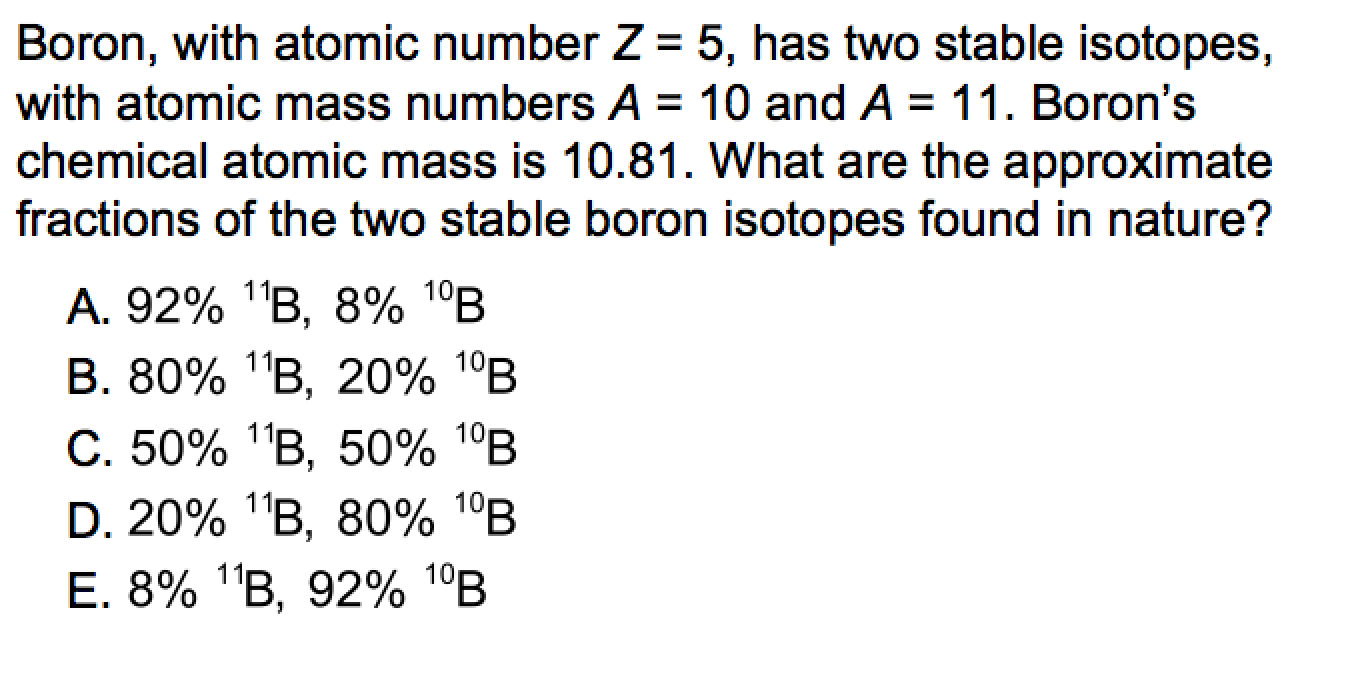
The mass number is written above the letter symbol. Boron Neutron Capture Therapy The fundamental concept of boron neutron. Element Boron (B), Group 13, Atomic Number 5, p-block, Mass 10.81. 
The mass number tells us the total number of protons and neutrons in an element. So, the atomic number for boron is 5, which means that boron will have 5 electrons (as well as 5 protons). This means that the atomic number for an element is the number of electrons as well as the number of protons. The atomic number for boron is 5, so boron will have 5 protons.Įlements are neutrally charged, which means that an element will have the same number of protons as electrons (remember, protons and electrons have the same magnitude of charge, but in opposite directions protons have a charge of +1 and electrons have a charge of -1). The atomic number tells us how many protons an element has, and it is written below the letter symbol.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
